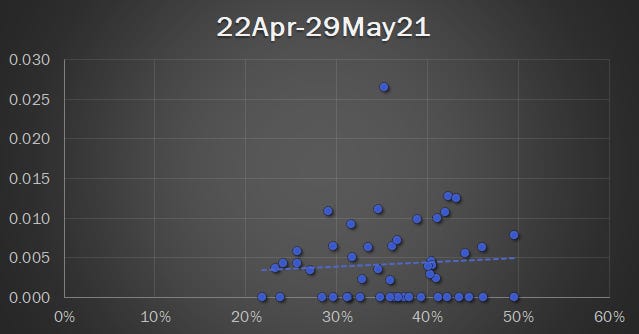
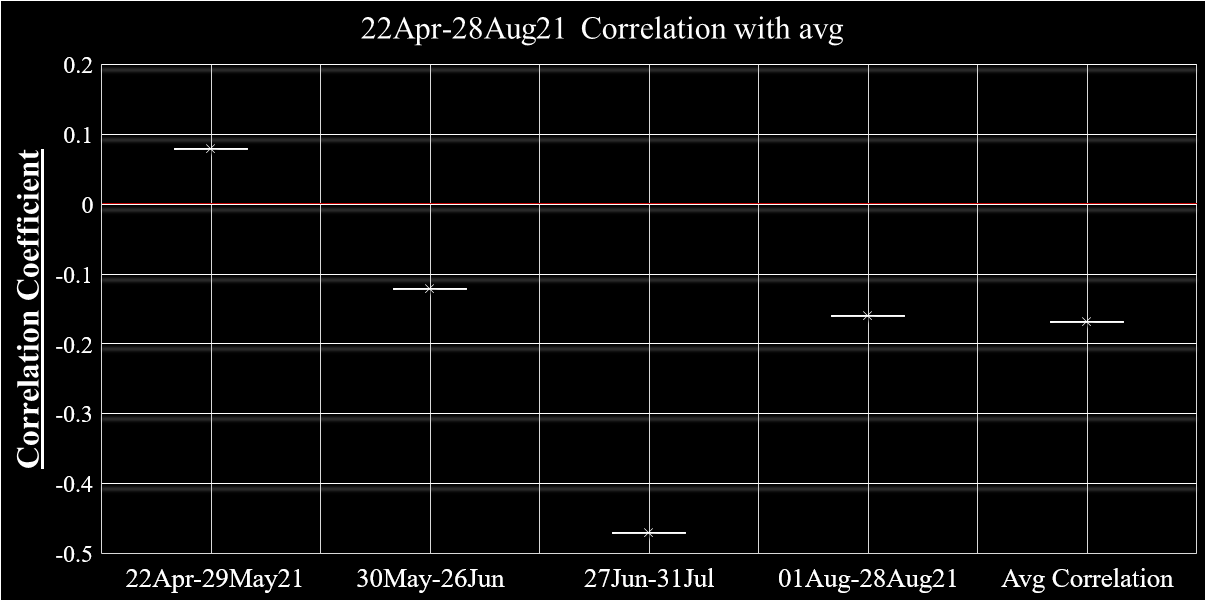
Datasets and graphics from Military Mandate Article
Below are datasets, charts, and references used throughout this write up.
caption...
Elflein, John. “Total Number of COVID-19 Cases by Age U.S. 2021.” Statista, October 8, 2021. https://www.statista.com/statistics/1254271/us-total-number-of-covid-cases-by-age-group/.
“CDC Wonder.” Centers for Disease Control and Prevention. Centers for Disease Control and Prevention. Accessed October 12, 2021. https://wonder.cdc.gov/.
“Provisional Deaths by Age and Sex Data.” Centers for Disease Control and Prevention. Centers for Disease Control and Prevention. Accessed October 12, 2021. https://data.cdc.gov/d/9bhg-hcku/visualization.
“CDC Covid Data Tracker Demographics.” Centers for Disease Control and Prevention. Centers for Disease Control and Prevention. Accessed October 12, 2021. https://covid.cdc.gov/covid-data-tracker/#demographics.
“Two More US Service Members Die from Covid-19, Bringing Military Deaths to 17.” American Military News, January 22, 2021. https://americanmilitarynews.com/2021/01/2-more-us-service-members-die-from-covid-19-bringing-military-deaths-to-17/.
18-49 age range used due to public health reporting. CDC and WHO data for cases and hospitalizations based on estimates with 95% UI*
“Coronavirus: Dod Response,” U.S. Department of Defense, accessed October 12, 2021, https://www.defense.gov/Spotlights/Coronavirus-DoD-Response/.
“Trends in Active-Duty Military Deaths since 2006.” Federation of American Scientists. Congressional Research Service. Accessed October 12, 2021. https://sgp.fas.org/crs/natsec/IF10899.pdf.
“Study to Describe the Safety, Tolerability, Immunogenicity, and Efficacy of RNA Vaccine Candidates against COVID-19 in Healthy Individuals - Full Text View.” Full Text View - ClinicalTrials.gov. Accessed October 12, 2021. https://clinicaltrials.gov/ct2/show/NCT04368728.
“Va.gov: Veterans Affairs,” How Long Before a Drug is Approved by the FDA? (US Department of Veteran Affairs, April 5, 2007), https://www.hiv.va.gov/patient/clinical-trials/drug-approval-process.asp.
“Drug Approval Process,” U.S. Food and Drug Administration (FDA Center for Drug Evaluation and Research), accessed October 9, 2021, https://www.fda.gov/media/83083/download.
Mulligan, Mark J., Kirsten E. Lyke, Nicholas Kitchin, Judith Absalon, Alejandra Gurtman, Stephen Lockhart, Kathleen Neuzil, et al. “Phase I/II Study of COVID-19 RNA Vaccine BNT162B1 in Adults.” Nature 586, no. 7830 (2020): 589–93. https://doi.org/10.1038/s41586-020-2639-4.
Polack, Fernando P., Stephen J. Thomas, Nicholas Kitchin, Judith Absalon, Alejandra Gurtman, Stephen Lockhart, John L. Perez, et al. “Safety and Efficacy of the BNT162B2 Mrna Covid-19 Vaccine.” New England Journal of Medicine 383, no. 27 (2020): 2603–15. https://doi.org/10.1056/nejmoa2034577.
Mark G. Thompson et al., “Prevention and Attenuation of Covid-19 with the BNT162B2 and MRNA-1273 Vaccines,” New England Journal of Medicine 385, no. 4 (2021): pp. 320-329, https://doi.org/10.1056/nejmoa2107058.
Thompson, Mark G., Jefferey L. Burgess, Allison L. Naleway, Harmony Tyner, Sarang K. Yoon, Jennifer Meece, Lauren E.W. Olsho, et al. “Prevention and Attenuation of Covid-19 with the BNT162B2 and MRNA-1273 Vaccines.” New England Journal of Medicine 385, no. 4 (2021): 320–29. https://doi.org/10.1056/nejmoa2107058.
“Pfizer and BioNTech Conclude Phase 3 Study of COVID-19 Vaccine Candidate, Meeting All Primary Efficacy Endpoints,” Pfizer (Pfizer-BioNTech, November 18, 2020), https://www.pfizer.com/news/press-release/press-release-detail/pfizer-and-biontech-conclude-phase-3-study-covid-19-vaccine.
Thomas, Stephen J., Edson D. Moreira, Nicholas Kitchin, Judith Absalon, Alejandra Gurtman, Stephen Lockhart, John L. Perez, et al. “Safety and Efficacy of the BNT162B2 Mrna Covid-19 Vaccine through 6 Months.” New England Journal of Medicine, 2021. https://doi.org/10.1056/nejmoa2110345.
“History of Changes for Study: NCT04368728 Study to Describe the Safety, Tolerability, Immunogenicity, and Efficacy of RNA Vaccine Candidates Against COVID-19 in Healthy Individuals.” History of changes for study: NCT04368728. Accessed October 12, 2021. https://clinicaltrials.gov/ct2/history/NCT04368728?V_32=View
“Pfizer and BioNTech Conclude Phase 3 Study of COVID-19 Vaccine Candidate, Meeting All Primary Efficacy Endpoints.” Pfizer. Pfizer-BioNTech, November 18, 2020. https://www.pfizer.com/news/press-release/press-release-detail/pfizer-and-biontech-conclude-phase-3-study-covid-19-vaccine.
Fernando P. Polack et al., “Safety and Efficacy of the BNT162B2 Mrna Covid-19 Vaccine,” New England Journal of Medicine 383, no. 27 (2020): pp. 2603-2615, https://doi.org/10.1056/nejmoa2034577.
“A Phase 1/2/3 Study to Evaluate the Safety, Tolerability, and Immunogenicity of an RNA Vaccine Candidate against COVID-19 in Healthy Children and Young Adults - Full Text View.” A Phase 1/2/3 Study to Evaluate the Safety, Tolerability, and Immunogenicity of an RNA Vaccine Candidate Against COVID-19 in Healthy Children and Young Adults. ClinicalTrials.gov. Accessed October 9, 2021.
“A Phase 1/2/3 Study to Evaluate the Safety, Tolerability, and Immunogenicity of an RNA Vaccine Candidate against COVID-19 in Healthy Children and Young Adults - Full Text View,” A Phase 1/2/3 Study to Evaluate the Safety, Tolerability, and Immunogenicity of an RNA Vaccine Candidate Against COVID-19 in Healthy Children and Young Adults (ClinicalTrials.gov), accessed October 9, 2021, https://clinicaltrials.gov/ct2/show/study/NCT04816643.
Ian Schwartz,On Date October 7, “Biden: The Vaccinated Are ‘Protected’ from Covid, ‘Cannot Spread It to You,’” RealClearPolitics (RealClear Politics, October 7, 2021), https://www.realclearpolitics.com/video/2021/10/07/biden_vaccinated_protected_from_covid_cannot_spread_it_to_you.html.
“CDC Director Changes Her Story, Now Admits Covid Vaccines Don’t Prevent Virus Transmission.” NOQ Report. Becker News, August 7, 2021. https://noqreport.com/2021/08/07/cdc-director-changes-her-story-now-admits-covid-vaccines-dont-prevent-virus-transmission/.
“US Centers for Disease Control,” US Centers for Disease Control (CDC, July 30, 2021), https://www.cdc.gov/media/releases/2021/s0730-mmwr-covid-19.html.
“Temporal Variation in Transmission during the Covid-19 Outbreak.” Covid-19 Temporal Variation in Transmission. Accessed October 12, 2021. https://epiforecasts.io/covid.
“Summary Data Reproductive Rate.” Harvard Dataverse, June 26, 2021. https://dataverse.harvard.edu/file.xhtml?fileId=4202836&version=8.0.
“State and National Provisional Counts.” Centers for Disease Control and Prevention. Centers for Disease Control and Prevention, October 6, 2021. https://www.cdc.gov/nchs/nvss/vsrr/provisional-tables.htm.
“United States Covid-19 Cases and Deaths by State over Time.” Centers for Disease Control and Prevention. Centers for Disease Control and Prevention. Accessed October 12, 2021. https://data.cdc.gov/Case-Surveillance/United-States-COVID-19-Cases-and-Deaths-by-State-o/9mfq-cb36.
Forgey, Quint. “Pentagon: 70 Percent of Service Members Have Received First Dose of Covid Vaccine.” POLITICO. POLITICO, July 18, 2021. https://www.politico.com/news/2021/07/16/military-coronavirus-vaccine-499822.
Ali, Zulfiqar, and S Bala Bhaskar. “Basic Statistical Tools in Research and Data Analysis.” Indian journal of anaesthesia. Medknow Publications & Media Pvt Ltd, September 2016. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5037948/.
Center for Biologics Evaluation and Research. “Influenza A (H1N1) 2009 Monovalent Vaccines Descriptions, Ingredients.” U.S. Food and Drug Administration. FDA. Accessed October 12, 2021. https://www.fda.gov/vaccines-blood-biologics/vaccines/influenza-h1n1-2009-monovalent-vaccines-descriptions-and-ingredients.
Forgey, Quint. “Pentagon: 70 Percent of Service Members Have Received First Dose of Covid Vaccine.” POLITICO. POLITICO, July 18, 2021. https://www.politico.com/news/2021/07/16/military-coronavirus-vaccine-499822.
“US Centers for Disease Control,” US Centers for Disease Control (CDC, July 30, 2021), https://www.cdc.gov/media/releases/2021/s0730-mmwr-covid-19.html.
Complete Reference list
Ali, Zulfiqar, and S Bala Bhaskar. “Basic Statistical Tools in Research and Data Analysis.” Indian journal of anaesthesia. Medknow Publications & Media Pvt Ltd, September 2016. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5037948/.
Becker, Kyle. “CDC Director Changes Her Story, Now Admits Covid Vaccines Don't Prevent Virus Transmission.” Becker News, August 7, 2021. https://beckernews.com/cdc-director-changes-her-story-now-admits-covid-vaccines-dont-prevent-virus-transmission-40754/.
Brown, Catherine M., Johanna Vostok, Hillary Johnson, Meagan Burns, Radhika Gharpure, Samira Sami, Rebecca T. Sabo, et al. “Outbreak of SARS-COV-2 Infections, Including COVID-19 Vaccine Breakthrough Infections, Associated with Large Public Gatherings — Barnstable County, Massachusetts, July 2021.” MMWR. Morbidity and Mortality Weekly Report 70, no. 31 (2021): 1059–62. https://doi.org/10.15585/mmwr.mm7031e2.
“CDC Covid Data Tracker Demographics.” Centers for Disease Control and Prevention. Centers for Disease Control and Prevention. Accessed October 12, 2021. https://covid.cdc.gov/covid-data-tracker/#demographics.
“CDC Director Changes Her Story, Now Admits Covid Vaccines Don't Prevent Virus Transmission.” NOQ Report. Becker News, August 7, 2021. https://noqreport.com/2021/08/07/cdc-director-changes-her-story-now-admits-covid-vaccines-dont-prevent-virus-transmission/.
“CDC Wonder.” Centers for Disease Control and Prevention. Centers for Disease Control and Prevention. Accessed October 12, 2021. https://wonder.cdc.gov/.
Center for Biologics Evaluation and Research. “Influenza A (H1N1) 2009 Monovalent Vaccines Descriptions, Ingredients.” U.S. Food and Drug Administration. FDA. Accessed October 12, 2021. https://www.fda.gov/vaccines-blood-biologics/vaccines/influenza-h1n1-2009-monovalent-vaccines-descriptions-and-ingredients.
“Coronavirus: Dod Response.” U.S. Department of Defense. Accessed October 12, 2021. https://www.defense.gov/Spotlights/Coronavirus-DOD-Response/.
“Coronavirus: Dod Response.” U.S. Department of Defense. Accessed October 12, 2021. https://www.defense.gov/Spotlights/Coronavirus-DoD-Response/.
“Covid Data Tracker Weekly Review.” Centers for Disease Control and Prevention. Centers for Disease Control and Prevention. Accessed October 12, 2021. https://www.cdc.gov/coronavirus/2019-ncov/covid-data/covidview/index.html.
“Covid-19: National and Subnational Estimates for the ...” National and Subnational estimates for the United States of America. EpiForecast, October 7, 2021. https://epiforecasts.io/covid/posts/national/united-states/.
“Drug Approval Process.” U.S. Food and Drug Administration. FDA Center for Drug Evaluation and Research. Accessed October 9, 2021. https://www.fda.gov/media/83083/download.
“Drug Safety Communication - FDA - Drug Approval.” Accessed October 12, 2021. https://www.fda.gov/media/86382/download.
Elflein, John. “Total Number of COVID-19 Cases by Age U.S. 2021.” Statista, October 8, 2021. https://www.statista.com/statistics/1254271/us-total-number-of-covid-cases-by-age-group/.
Feldman, Robert A., Rainard Fuhr, Igor Smolenov, Amilcar (Mick) Ribeiro, Lori Panther, Mike Watson, Joseph J. Senn, et al. “MRNA Vaccines against H10N8 and H7N9 Influenza Viruses of Pandemic Potential Are Immunogenic and Well Tolerated in Healthy Adults in Phase 1 Randomized Clinical Trials.” Vaccine 37, no. 25 (2019): 3326–34. https://doi.org/10.1016/j.vaccine.2019.04.074.
Fitzsimons, Tim. “California to Require Covid Vaccine for Schoolchildren, Newsom Announces.” NBCNews.com. NBCUniversal News Group, October 1, 2021. https://www.nbcnews.com/news/us-news/california-require-covid-vaccine-public-schoolchildren-newsom-announces-n1280602.
Forgey, Quint. “Pentagon: 70 Percent of Service Members Have Received First Dose of Covid Vaccine.” POLITICO. POLITICO, July 18, 2021. https://www.politico.com/news/2021/07/16/military-coronavirus-vaccine-499822.
“History of Changes for Study: NCT04368728 Study to Describe the Safety, Tolerability, Immunogenicity, and Efficacy of RNA Vaccine Candidates Against COVID-19 in Healthy Individuals.” History of changes for study: NCT04368728. Accessed October 12, 2021. https://clinicaltrials.gov/ct2/history/NCT04368728?V_32=View.
“Influenza A (H1N1) 2009 Monovalent Vaccine (H1N1 Influenza Virus Vaccine) FDA Approval History.” Drugs.com. Accessed October 12, 2021. https://www.drugs.com/history/influenza-a-h1n1-2009-monovalent-vaccine.html.
Mukaka, M M. “Statistics Corner: A Guide to Appropriate Use of Correlation Coefficient in Medical Research.” Malawi medical journal : the journal of Medical Association of Malawi. The Medical Association Of Malawi, September 2012. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3576830/.
Mulligan, Mark J., Kirsten E. Lyke, Nicholas Kitchin, Judith Absalon, Alejandra Gurtman, Stephen Lockhart, Kathleen Neuzil, et al. “Phase I/II Study of COVID-19 RNA Vaccine BNT162B1 in Adults.” Nature 586, no. 7830 (2020): 589–93. https://doi.org/10.1038/s41586-020-2639-4.
“Pfizer and BioNTech Conclude Phase 3 Study of COVID-19 Vaccine Candidate, Meeting All Primary Efficacy Endpoints.” Pfizer. Pfizer-BioNTech, November 18, 2020. https://www.pfizer.com/news/press-release/press-release-detail/pfizer-and-biontech-conclude-phase-3-study-covid-19-vaccine.
“A Phase 1/2/3 Study to Evaluate the Safety, Tolerability, and Immunogenicity of an RNA Vaccine Candidate against COVID-19 in Healthy Children and Young Adults - Full Text View.” A Phase 1/2/3 Study to Evaluate the Safety, Tolerability, and Immunogenicity of an RNA Vaccine Candidate Against COVID-19 in Healthy Children and Young Adults. ClinicalTrials.gov. Accessed October 9, 2021. https://clinicaltrials.gov/ct2/show/study/NCT04816643.
“A Phase 1/2/3 Study to Evaluate the Safety, Tolerability, and Immunogenicity of an RNA Vaccine Candidate against COVID-19 in Healthy Children and Young Adults - Full Text View.” Full Text View - ClinicalTrials.gov. Accessed October 12, 2021. https://clinicaltrials.gov/ct2/show/study/NCT04816643.
Polack, Fernando P., Stephen J. Thomas, Nicholas Kitchin, Judith Absalon, Alejandra Gurtman, Stephen Lockhart, John L. Perez, et al. “Safety and Efficacy of the BNT162B2 Mrna Covid-19 Vaccine.” New England Journal of Medicine 383, no. 27 (2020): 2603–15. https://doi.org/10.1056/nejmoa2034577.
Polack, Fernando P., Stephen J. Thomas, Nicholas Kitchin, Judith Absalon, Alejandra Gurtman, Stephen Lockhart, John L. Perez, et al. “Safety and Efficacy of the BNT162B2 Mrna Covid-19 Vaccine.” New England Journal of Medicine 383, no. 27 (2020): 2603–15. https://doi.org/10.1056/nejmoa2034577.
Polack, Fernando P., Stephen J. Thomas, Nicholas Kitchin, Judith Absalon, Alejandra Gurtman, Stephen Lockhart, John L. Perez, et al. “Safety and Efficacy of the BNT162B2 Mrna Covid-19 Vaccine.” New England Journal of Medicine 383, no. 27 (2020): 2603–15. https://doi.org/10.1056/nejmoa2034577.
“Provisional Deaths by Age and Sex Data.” Centers for Disease Control and Prevention. Centers for Disease Control and Prevention. Accessed October 12, 2021. https://data.cdc.gov/d/9bhg-hcku/visualization.
Ritchie, Hannah, Edouard Mathieu, Lucas Rodés-Guirao, Cameron Appel, Charlie Giattino, Esteban Ortiz-Ospina, Joe Hasell, Bobbie Macdonald, Diana Beltekian, and Max Roser. “Coronavirus (COVID-19) Hospitalizations - Statistics and Research.” Our World in Data, March 5, 2020. https://ourworldindata.org/covid-hospitalizations.
Schwartz On Date October 7, Ian. “Biden: The Vaccinated Are ‘Protected’ from Covid, ‘Cannot Spread It to You.’” RealClearPolitics. RealClear Politics, October 7, 2021. https://www.realclearpolitics.com/video/2021/10/07/biden_vaccinated_protected_from_covid_cannot_spread_it_to_you.html.
“State and National Provisional Counts.” Centers for Disease Control and Prevention. Centers for Disease Control and Prevention, October 6, 2021. https://www.cdc.gov/nchs/nvss/vsrr/provisional-tables.htm.
“Statement from CDC Director Rochelle P. Walensky, MD, MPH on Today’s MMWR.” US Centers for Disease Control. CDC, July 30, 2021. CDC. https://www.cdc.gov/media/releases/2021/s0730-mmwr-covid-19.html.
“Study to Describe the Safety, Tolerability, Immunogenicity, and Efficacy of RNA Vaccine Candidates against COVID-19 in Healthy Individuals - Full Text View.” Full Text View - ClinicalTrials.gov. Accessed October 12, 2021. https://clinicaltrials.gov/ct2/show/NCT04368728.
“Summary Data Reproductive Rate.” Harvard Dataverse, June 26, 2021. https://dataverse.harvard.edu/file.xhtml?fileId=4202836&version=8.0.
“Temporal Variation in Transmission during the Covid-19 Outbreak.” Covid-19 Temporal Variation in Transmission. Accessed October 12, 2021. https://epiforecasts.io/covid.
Thomas, Stephen J., Edson D. Moreira, Nicholas Kitchin, Judith Absalon, Alejandra Gurtman, Stephen Lockhart, John L. Perez, et al. “Safety and Efficacy of the BNT162B2 Mrna Covid-19 Vaccine through 6 Months.” New England Journal of Medicine, 2021. https://doi.org/10.1056/nejmoa2110345.
Thompson, Mark G., Jefferey L. Burgess, Allison L. Naleway, Harmony Tyner, Sarang K. Yoon, Jennifer Meece, Lauren E.W. Olsho, et al. “Prevention and Attenuation of Covid-19 with the BNT162B2 and MRNA-1273 Vaccines.” New England Journal of Medicine 385, no. 4 (2021): 320–29. https://doi.org/10.1056/nejmoa2107058.
Treatment. “Va.gov: Veterans Affairs.” How Long Before a Drug is Approved by the FDA?, April 5, 2007. https://www.hiv.va.gov/patient/clinical-trials/drug-approval-process.asp.
“Trends in Active-Duty Military Deaths since 2006.” Federation of American Scientists. Congressional Research Service. Accessed October 12, 2021. https://sgp.fas.org/crs/natsec/IF10899.pdf.
“Two More US Service Members Die from Covid-19, Bringing Military Deaths to 17.” American Military News, January 22, 2021. https://americanmilitarynews.com/2021/01/2-more-us-service-members-die-from-covid-19-bringing-military-deaths-to-17/.
“United States Covid-19 Cases and Deaths by State over Time.” Centers for Disease Control and Prevention. Centers for Disease Control and Prevention. Accessed October 12, 2021. https://data.cdc.gov/Case-Surveillance/United-States-COVID-19-Cases-and-Deaths-by-State-o/9mfq-cb36.
Uria, Daniel, and Clyde Hughes. “Biden Calls on Unvaccinated to 'Do the Right Thing' While Touting Mandate in Chicago.” UPI. UPI, October 7, 2021. https://www.upi.com/Top_News/US/2021/10/07/joe-biden-vaccine-mandates-chicago/7301633602898/.
“Va.gov: Veterans Affairs.” How Long Before a Drug is Approved by the FDA? US Department of Veteran Affairs, April 5, 2007. https://www.hiv.va.gov/patient/clinical-trials/drug-approval-process.asp.
Vogel, Annette B., Isis Kanevsky, Ye Che, Kena A. Swanson, Alexander Muik, Mathias Vormehr, Lena M. Kranz, et al. “BNT162B Vaccines Protect Rhesus Macaques from SARS-COV-2.” Nature 592, no. 7853 (2021): 283–89. https://doi.org/10.1038/s41586-021-03275-y.
Walsh, Edward E., Robert Frenck, Ann R. Falsey, Nicholas Kitchin, Judith Absalon, Alejandra Gurtman, Stephen Lockhart, et al. “RNA-Based COVID-19 Vaccine BNT162b2 Selected for a Pivotal Efficacy Study,” 2020. https://doi.org/10.1101/2020.08.17.20176651.
Walsh, Edward E., Robert W. Frenck, Ann R. Falsey, Nicholas Kitchin, Judith Absalon, Alejandra Gurtman, Stephen Lockhart, et al. “Safety and Immunogenicity of Two RNA-Based Covid-19 Vaccine Candidates.” New England Journal of Medicine 383, no. 25 (2020): 2439–50. https://doi.org/10.1056/nejmoa2027906.